The topicity relationship of \(H_a\) and \(H_b\) in X, Y and Z (as drawn in the figure) are, respectively,

The topicity relationship of \(H_a\) and \(H_b\) in X, Y and Z (as drawn in the figure) are, respectively, 
Show Hint
- Diastereotopic, Homotopic and Enantiotopic
- Homotopic, Enantiotopic and Enantiotopic
- Homotopic, Homotopic and Enantiotopic
- Diastereotopic, Enantiotopic and Homotopic
The Correct Option is B
Solution and Explanation
Step 1: Molecule X. The two hydrogens \(H_a\) and \(H_b\) lie on a carbon in a highly symmetric (meso/C\(_2\)) environment bearing identical HO− and −CO2H substituent sets on opposite sides. A \(C_2\) rotation superposes \(H_a\) and \(H_b\); replacing either gives the same molecule \(⇒\) homotopic.
Step 2: Molecule Y. In the bridged/aromatic framework, \(H_a\) and \(H_b\) are related by an improper/mirror operation: replacement of \(H_a\) versus \(H_b\) generates non-superposable mirror-image products, while the parent is achiral. Thus they are enantiotopic.
Step 3: Molecule Z. For the prochiral vinyl \({CH_2}\) adjacent to −Cl, the two vinylic hydrogens are in an enantiotopic relationship (replacing one or the other creates enantiomeric \(E/R_e\) vs \(S_i\) labeled products). Hence enantiotopic.
Step 4: Collecting: \(X\) homotopic, \(Y\) enantiotopic, \(Z\) enantiotopic \(⇒\) option (B).
Top GATE CY Organic Chemistry Questions
- The major product in the following reaction sequence is
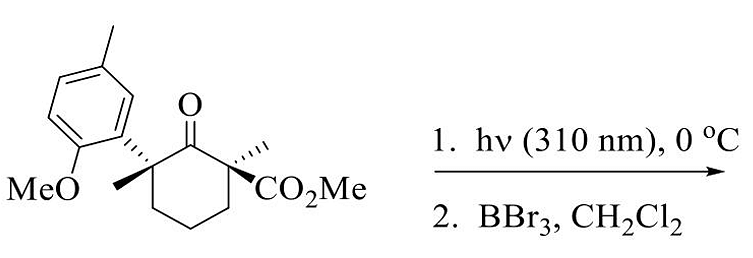
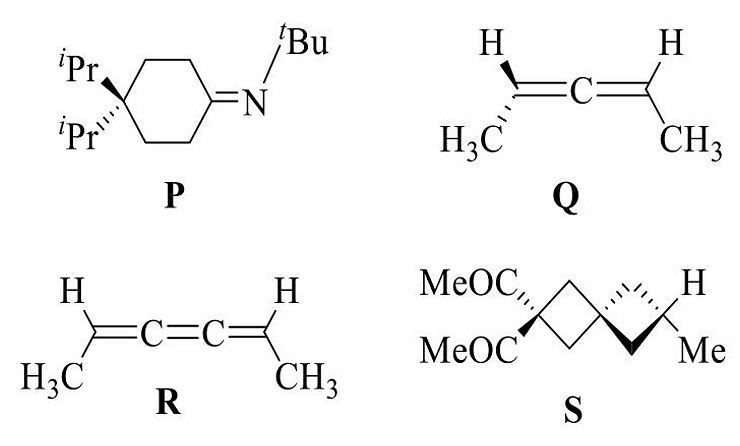
- Among the following, the chiral compound is
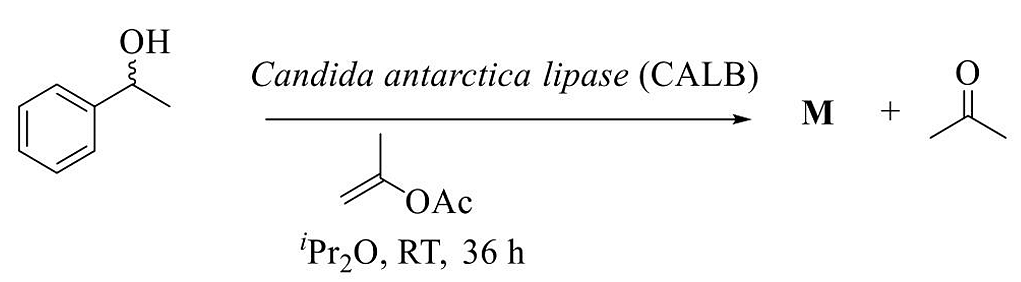
- The product M in the following reaction is
- The reaction(s) that yield(s) X as the major product is (are)
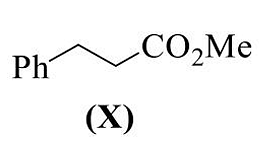
- The reaction(s) that yield(s) 2-methylquinoline as the major product is (are)
Top GATE CY Relative stereochemistry in compounds having more than one stereogenic centre Questions
- The correct statement(s) for decalin is (are)
- Fischer presentation of D-(-)-fructose is given below.
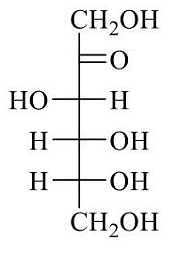
The correct structure of α-L-(+)-fructofuranose is - The major products X and Y in the following reaction sequence are
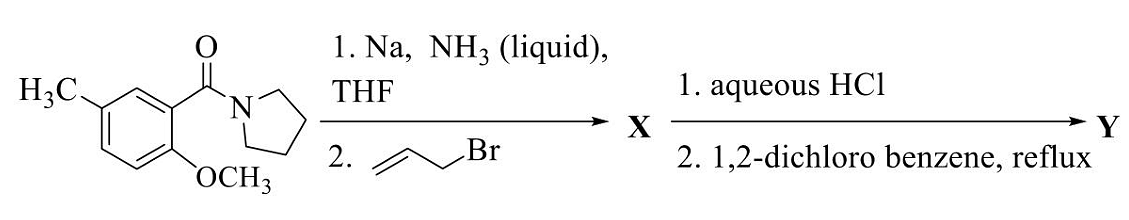
The acetolysis product(s) of the given reaction (solvolysis of the benzylic tosylate in AcOH) is(are)

The reaction(s) in which inversion of configuration occur(s) is(are)

Top GATE CY Questions
- Ψ(x) = \(\sqrt{\frac{32}{L^3}}\)[sin(\(\frac{\pi x}{L}\)).cos(\(\frac{\pi x}{L}\)).sin(\(\frac{2\pi y}{L}\)).sin(\(\frac{\pi z}{L}\))]Find the value of E221: E111.
 what is the final product
what is the final product- Which are optically active?
 intensity ratio of final product
intensity ratio of final product- Which among the following is true?