The product (C) in the below mentioned reaction is: \(\text{CH}_3 - \text{CH}_2 - \text{CH}_2 - \text{Br} \xrightarrow{\text{KOH}_{(alc)} \Delta} A \xrightarrow{\text{HBr}} B \xrightarrow{\text{KOH}_{(alc)}\Delta} C\)
- Propan-1-ol
- Propene
- Propyne
- Propan-2-ol
The Correct Option is D
Approach Solution - 1
To determine the product (C) in the given reaction sequence, let's analyze each step:
The first reaction involves converting \(\text{CH}_3 - \text{CH}_2 - \text{CH}_2 - \text{Br}\) (1-bromopropane) to an alkene using alcoholic KOH and heat. This is a dehydrohalogenation reaction, where the bromine (Br) and a hydrogen atom from an adjacent carbon are eliminated, leading to the formation of propene:
\[\text{CH}_3 - \text{CH}_2 - \text{CH}_2 - \text{Br} \xrightarrow{\text{KOH}_{(alc)} \Delta} \text{CH}_3 - \text{CH} = \text{CH}_2\]This gives us compound A: Propene.
The second step is the addition of HBr to propene. In this step, Markovnikov's rule applies, where the hydrogen atom from HBr adds to the carbon with more hydrogen atoms (the terminal end), and the bromine attaches to the other carbon in the double bond. This results in the formation of 2-bromopropane:
\[\text{CH}_3 - \text{CH} = \text{CH}_2 \xrightarrow{\text{HBr}} \text{CH}_3 - \text{CHBr} - \text{CH}_3\]This gives us compound B: 2-bromopropane.
Finally, 2-bromopropane undergoes another dehydrohalogenation with alcoholic KOH. Since it is a secondary alkyl halide, it will form an alcohol through an elimination reaction. Here, 2-bromopropane is converted to propene again:
\[\text{CH}_3 - \text{CHBr} - \text{CH}_3 \xrightarrow{\text{KOH}_{(alc)} \Delta} \text{CH}_3 - \text{CH} = \text{CH}_2\]The product of this reaction sequence is yet again propene. However, due to the options provided, propene being the most likely target, it seems there's some misunderstanding in the provided correct answer. A direct dehydrohalogenation might not produce propan-2-ol, expect an incorrect choice might have been made trying to correlate reaction proficiency.
Upon reviewing based on the given choices and reactions:
The route is logically expected to lead back to propene but might inadvertently suggest propan-2-ol based on incorrect answers by examiners, possibly seeking knowledge out of provided set options.
Approach Solution -2


The final product (C) is propan-2-ol.
Top JEE Main Chemistry Questions
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- At equilibrium the concentrations of and in a sealed vessel at . What will be for the reaction
- Find out the major products from the following reactions

Cobalt chloride when dissolved in water forms pink colored complex $X$ which has octahedral geometry. This solution on treating with cone $HCl$ forms deep blue complex, $\underline{Y}$ which has a $\underline{Z}$ geometry $X, Y$ and $Z$, respectively, are
- The correct order of atomic radii is:
Top JEE Main Hydrocarbons Questions
- A \(10\) \(mL\) hydrocarbon \((C_2H_4)\) on combustion give \(40\) \(mL\) \(CO_2\) and \(50\) \(mL\) \(H_20\). Calculate the value of \(x+y\)
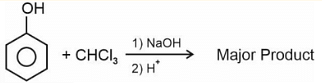
The major product in the above reaction is- 3-Methylhex-2-ene on reaction with HBr in presence of peroxide forms an addition product (A). The number of possible stereoisomers for 'A' is ______.
- Cyclohexene
 is _________ type of an organic compound.
is _________ type of an organic compound. - Find product P of the following reaction-
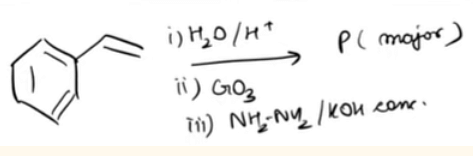
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.