The number of chlorine atoms in bithionol is ______ .
Correct Answer: 4
Approach Solution - 1
Bithionol is a chemical compound used as an anthelmintic agent and a disinfectant. Its chemical formula is C12H6Cl4O2S. To determine the number of chlorine atoms in bithionol, we examine its chemical formula.
The subscript '4' following the 'Cl' symbol in C12H6Cl4O2S indicates there are four chlorine atoms.
Thus, the number of chlorine atoms in bithionol is 4.
Confirming this against the provided range (4,4), we see that the calculated value fits perfectly within the range.
Approach Solution -2

Number of chlorine atoms in bithionol = 4
So, the answer is 4.
Top JEE Main Chemistry Questions
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- At equilibrium the concentrations of and in a sealed vessel at . What will be for the reaction
- Find out the major products from the following reactions

Cobalt chloride when dissolved in water forms pink colored complex $X$ which has octahedral geometry. This solution on treating with cone $HCl$ forms deep blue complex, $\underline{Y}$ which has a $\underline{Z}$ geometry $X, Y$ and $Z$, respectively, are
- The correct order of atomic radii is:
Top JEE Main Hydrocarbons Questions
- A \(10\) \(mL\) hydrocarbon \((C_2H_4)\) on combustion give \(40\) \(mL\) \(CO_2\) and \(50\) \(mL\) \(H_20\). Calculate the value of \(x+y\)
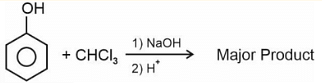
The major product in the above reaction is- 3-Methylhex-2-ene on reaction with HBr in presence of peroxide forms an addition product (A). The number of possible stereoisomers for 'A' is ______.
- Cyclohexene
 is _________ type of an organic compound.
is _________ type of an organic compound. - Find product P of the following reaction-
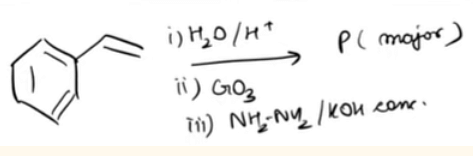
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.
Concepts Used:
Hydrocarbons
Hydrocarbons can be described as organic compounds that consists only hydrogen and carbon atoms. These compounds are of different types and thereby have distinct natures. Hydrocarbons are colorless gases and are known for discharging faint odours. These have been categorized under four major classes named as alkynes, alkanes, alkenes, and aromatic hydrocarbons.
Types of Hydrocarbons
- Saturated hydrocarbons - Saturated hydrocarbons are those compounds where there is a single bond exists between carbon atoms and are saturated with atoms of hydrogen.
- Unsaturated hydrocarbons - Hydrocarbons comprises of at least one double or triple bond between carbon atoms are known as unsaturated hydrocarbons.
- Aliphatic hydrocarbons - The term denotes the hydrocarbons formed as an outcome of the chemical degradation of fats. Aliphatic hydrocarbons are basically chemical compounds.
- Aromatic hydrocarbons - They are distinguished because of the presence of benzene rings in them. They give away distinct types of aroma. These hydrocarbons comprises of only hydrogen and carbon atoms.