The alkane from below having two secondary hydrogens is:
Show Hint
- 2,2,4,4-Tetramethylhexane
- 2,2,3-Tetramethylpentane
- 4-Ethyl-3,4-dimethyloctane
- 2,2,4,5-Tetramethylheptane
The Correct Option is B
Solution and Explanation
Step 1: A secondary hydrogen is one that is attached to a carbon atom that is itself attached to two other carbon atoms.
Step 2: In 2,2,3-Tetramethylpentane, the carbon atoms at positions 2 and 3 have two other carbon atoms attached, and the hydrogens attached to these carbons are secondary hydrogens.
Step 3: Other options do not exhibit the same secondary hydrogen placement.
Thus, the correct answer is 2,2,3-Tetramethylpentane.
Top JEE Main Chemistry Questions
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- At equilibrium the concentrations of and in a sealed vessel at . What will be for the reaction
- Find out the major products from the following reactions

Cobalt chloride when dissolved in water forms pink colored complex $X$ which has octahedral geometry. This solution on treating with cone $HCl$ forms deep blue complex, $\underline{Y}$ which has a $\underline{Z}$ geometry $X, Y$ and $Z$, respectively, are
- The correct order of atomic radii is:
Top JEE Main Hydrocarbons Questions
- A \(10\) \(mL\) hydrocarbon \((C_2H_4)\) on combustion give \(40\) \(mL\) \(CO_2\) and \(50\) \(mL\) \(H_20\). Calculate the value of \(x+y\)
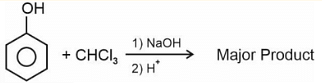
The major product in the above reaction is- 3-Methylhex-2-ene on reaction with HBr in presence of peroxide forms an addition product (A). The number of possible stereoisomers for 'A' is ______.
- Cyclohexene
 is _________ type of an organic compound.
is _________ type of an organic compound. - Find product P of the following reaction-
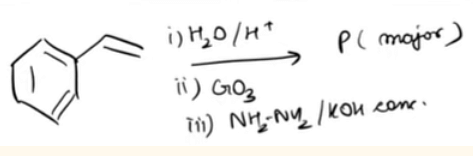
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.