Given below are two statements:
Statement I : In froth floatation method a rotating paddle agitates the mixture to drive air out of it.
Statement II : Iron pyrites are generally avoided for extraction of iron due to environmental reasons.
In the light of the above statements, choose the correct answer from the options given below:
Statement I : In froth floatation method a rotating paddle agitates the mixture to drive air out of it.
Statement II : Iron pyrites are generally avoided for extraction of iron due to environmental reasons.
In the light of the above statements, choose the correct answer from the options given below:
- Both Statement I and Statement II are false
- Both Statement I and Statement II are true
- Statement I is false but Statement II is true
- Statement I is true but Statement II is false
The Correct Option is C
Solution and Explanation
Statement I is false. In froth flotation, the primary purpose of the rotating paddle is to introduce air into the mixture, not to drive it out. The paddle helps create bubbles that attach to the mineral particles, making them float to the surface, where they can be collected as froth. This process is essential for the separation of valuable minerals from the gangue in the ore. Therefore, the statement that the paddle drives air out is incorrect.
Statement II is true. Iron pyrites (Fe\(S_2\)), when exposed to air and water, undergo oxidation and produce sulfuric acid. This acidic runoff is known as acid mine drainage (AMD), which can lead to severe environmental harm by contaminating water sources and soils. The acid dissolves heavy metals like copper and lead from surrounding rocks, further contributing to pollution. Hence, the statement about iron pyrites leading to acidic runoff is accurate.
Top JEE Main Chemistry Questions
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- At equilibrium the concentrations of and in a sealed vessel at . What will be for the reaction
- Find out the major products from the following reactions

Cobalt chloride when dissolved in water forms pink colored complex $X$ which has octahedral geometry. This solution on treating with cone $HCl$ forms deep blue complex, $\underline{Y}$ which has a $\underline{Z}$ geometry $X, Y$ and $Z$, respectively, are
- The correct order of atomic radii is:
Top JEE Main Concentration of ores Questions
The methods NOT involved in concentration of ore are
A Liquation
B Leaching
C Electrolysis
D Hydraulic washing
E Froth floatation
Choose the correct answer from the options given below :- Among the following ores Bauxite, Siderite, Cuprite, Calamine, Haematite, Kaolinite, Malachite, Magnetite, Sphalerite, Limonite, Cryolite, the number of principal ores of iron is____.
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.
Concepts Used:
General Principles and Processes of Isolation of Elements
What are Ores and Minerals?
Minerals are the naturally occurring, homogeneous inorganic solid substances. They are having a definite chemical composition and crystalline structure, hardness and color. For example, copper pyrite, calamine, etc.
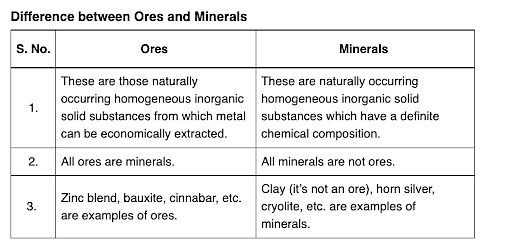
Impurities in an ore are called gauge. The removal of a gauge from the ore is called concentration ore.
Several steps are involved in the extraction of pure metal from ores. Major steps are as follows –
- Concentration of the ore
- Isolation of the metal from its concentrated ore
- Purification of the metal