Potential energy of two diatomic molecules P and Q of the same reduced mass is shown in the figure. According to this diagram, which of the following option(s) is/are correct?
Show Hint
- The equilibrium inter-nuclear distance of Q is more than that of P
- The total energy \( E = 0 \) separates bound and unbound states of the molecules
- The lowest vibrational frequency of P is larger than that of Q
- Dissociation energy of Q is more than that of P
The Correct Option is A, B, C
Solution and Explanation
The question presents a potential energy diagram for two diatomic molecules, P and Q. Let's analyze each option based on the provided diagram:
-
Equilibrium Inter-Nuclear Distance:
The equilibrium inter-nuclear distance corresponds to the minimum of the potential energy curve. From the diagram, the equilibrium inter-nuclear distance for molecule Q is larger than that for molecule P.
✓ Option (A) is correct.
-
Energy E = 0 Separates Bound and Unbound States:
The energy level E = 0 represents the threshold between bound and unbound molecular states. States with energy below zero are bound (stable), and those above zero are unbound (dissociative).
✓ Option (B) is correct.
-
Lowest Vibrational Frequency:
Vibrational frequency is linked to the curvature of the potential energy curve at the minimum. A steeper curve (as in P) indicates stronger restoring forces and thus a higher vibrational frequency compared to a shallower curve (as in Q).
✓ Option (C) is correct.
-
Dissociation Energy:
Dissociation energy is the depth of the potential well. From the diagram, molecule Q has a deeper well, hence a higher dissociation energy than P.
✗ Option (D) is incorrect.
Conclusion: The correct options are (A), (B), and (C).
Top GATE PH Physics Questions
- An infinite one-dimensional lattice extends along the x-axis. At each lattice site, there exists an ion with spin 1/2. The spin can point either in the +z or -z direction only. Let \( S_P \), \( S_F \), and \( S_A \) denote the entropies of paramagnetic, ferromagnetic, and antiferromagnetic configurations, respectively. Which of the following relations is/are true?
- Two point charges of charge \( +q \) each are placed a distance \( 2d \) apart. A grounded solid conducting sphere of radius \( a \) is placed midway between them. Assume \( a^2 \ll d^2 \). Which of the following statements is/are true?
- A particle of mass $m$ is moving in the potential
\[ V(x) = \begin{cases} V_0 + \frac{1}{2} m \omega_{0P}^2 x^2 & \quad \text{if } x > 0 \\ \infty & \quad \text{if } x \leq 0 \end{cases} \]
Figures P, Q, R, and S show different combinations of the values of $ \omega_0 $ and $ V_0 $. - Decays of mesons and baryons can be categorized as weak, strong and electromagnetic decays depending upon the interactions involved in the processes. Which of the following option is/are true?
- The temperature \( T \) dependence of magnetic susceptibility \( \chi \) (Column I) of certain magnetic materials (Column II) are given below. Which of the following options is/are correct?
Column I Column II (1) 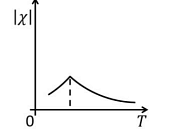
(P) Diamagnetic (2) 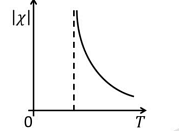
(Q) Paramagnetic (3) 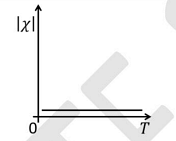
(R) Ferromagnetic (4) 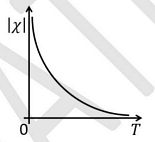
(S) Antiferromagnetic
Top GATE PH Questions
- If \( F_1(Q, q) = Qq \) is the generating function of a canonical transformation from \((p, q)\) to \((P, Q)\), then which one of the following relations is correct?
- When an unpolarized plane electromagnetic wave in a dielectric medium with refractive index \( n_1 \) is incident on a plane interface separating it from another dielectric medium with refractive index \( n_2 \) (where \( n_2 > n_1 \)), and the angle of incidence is \(\tan^{-1} \left( \frac{n_2}{n_1} \right) \), the following statement is true:
- The wavefunction of a particle in an infinite one-dimensional potential well at time \( t \) is
\[ \Psi(x, t) = \sqrt{\frac{2}{3}} e^{-iE_1 t/\hbar}\psi_1(x) + \frac{1}{\sqrt{6}} e^{i\pi/6} e^{-iE_2 t/\hbar} \psi_2(x) + \frac{1}{\sqrt{6}} e^{i\pi/4} e^{-iE_3 t/\hbar} \psi_3(x) \]where \(\psi_1\), \(\psi_2\), and \(\psi_3\) are the normalized ground state, the normalized first excited state, and the normalized second excited state, respectively. \(E_1\), \(E_2\), and \(E_3\) are the eigen-energies corresponding to \(\psi_1\), \(\psi_2\), and \(\psi_3\), respectively. The expectation value of energy of the particle in state \(\Psi(x,t)\) is - If a thermodynamical system is adiabatically isolated and experiences a change in volume under an externally applied constant pressure, then the thermodynamical potential minimized at equilibrium is the
- The mean distance between the two atoms of an HD molecule is \( r \), where H and D denote hydrogen and deuterium, respectively. The mass of the hydrogen atom is \( m_H \). The energy difference between the two lowest lying rotational states of HD in multiples of \(\frac{h^2}{m_H r^2}\) is: