Question:
Plane P3 is passing through (1,1,1) and line of intersection of P1 and P2 where \(P_{1}: 2x - y + z = 5\) and \(P_{2}: x + 3y + 2z + 2 = 0\). Then distance of (1,1,10) from P3 is:
Plane P3 is passing through (1,1,1) and line of intersection of P1 and P2 where \(P_{1}: 2x - y + z = 5\) and \(P_{2}: x + 3y + 2z + 2 = 0\). Then distance of (1,1,10) from P3 is:
Updated On: Mar 29, 2026
- \(\frac{53}{85}\)
\(\sqrt{85}\)
\(\frac{63}{\sqrt{85}}\)
- 53
Show Solution
Verified By Collegedunia
The Correct Option is C
Solution and Explanation
The correct option is (C): \(\frac{52}{\sqrt{85}}\)
Was this answer helpful?
0
0
Top JEE Main Mathematics Questions
- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.
- If , then the general value of is:
- The general solution of
- If the constant term in the binomial expansion of $\left(\frac{x^{\frac{3}{2}}}{2}-\frac{4}{x^l}\right)^9$ is $-84$ and the coefficient of $x^{-3 l}$ is $2^\alpha \beta$, where $\beta<0$ is an odd number, then $|\alpha l-\beta|$ is equal to_____
View More Questions
Top JEE Main Three Dimensional Geometry Questions
- If the foot is perpendicular from (1, 2, 3) to the line \(\frac{x+1}{2} = \frac{y-2}{5} = \frac{z-1}{1}\) is \(( a, \beta, \gamma)\), then find \(a + \beta + \gamma\)
- The sum of all values of $\alpha$, for which the shortest distance between the lines $\dfrac{x+1}{\alpha}=\dfrac{y-2}{-1}=\dfrac{z-4}{-\alpha}$ and $\dfrac{x}{\alpha}=\dfrac{y-1}{2}=\dfrac{z-1}{2\alpha}$ is $\sqrt{2}$, is
- If the distances of the point \( (1,2,a) \) from the line \[ \frac{x-1}{1}=\frac{y}{2}=\frac{z-1}{1} \] along the lines \[ L_1:\ \frac{x-1}{3}=\frac{y-2}{4}=\frac{z-a}{b} \quad \text{and} \quad L_2:\ \frac{x-1}{1}=\frac{y-2}{4}=\frac{z-a}{c} \] are equal, then \( a+b+c \) is equal to:
- The value of the integral \( \int_{\frac{\pi}{24}}^{\frac{5\pi}{24}} \frac{dx}{1 + \sqrt[3]{\tan 2x}} \) is :
- If $\lambda_1<\lambda_2$ are two values of $\lambda$ such that the angle between the planes $P_1: \vec{r}(3 \hat{i}-5 \hat{j}+\hat{k})=7$ and $P_2: \vec{r} \cdot(\lambda \hat{i}+\hat{j}-3 \hat{k})=9$ is $\sin ^{-1}\left(\frac{2 \sqrt{6}}{5}\right)$, then the square of the length of perpendicular from the point $\left(38 \lambda_1, 10 \lambda_2, 2\right)$ to the plane $P_1$ is _____
View More Questions
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.
View More Questions
Concepts Used:
Distance of a Point from a Plane
The shortest perpendicular distance from the point to the given plane is the distance between point and plane. In simple terms, the shortest distance from a point to a plane is the length of the perpendicular parallel to the normal vector dropped from the particular point to the particular plane. Let's see the formula for the distance between point and plane.
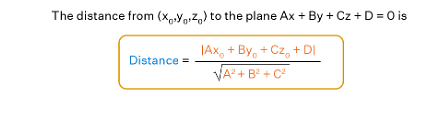
Read More: Distance Between Two Points