Question:
In the removal of silica during smelting, the flux used is
In the removal of silica during smelting, the flux used is
Updated On: Apr 1, 2026
- ferric oxide
- calcium oxide
- cupric oxide
- phosphorus pentoxide
Show Solution
Verified By Collegedunia
The Correct Option is B
Solution and Explanation
Calcium oxide acts as a flux and combines with silica to form fusible calcium silicate slag
$ {CaO_{(s)} + SiO_{2(s)} ->[1123 K] $\underset{\text{Calcium silicate (slag)}}{ {CaSiO_{3(s)} }}$ }$
$ {CaO_{(s)} + SiO_{2(s)} ->[1123 K] $\underset{\text{Calcium silicate (slag)}}{ {CaSiO_{3(s)} }}$ }$
Was this answer helpful?
0
0
Top COMEDK UGET Chemistry Questions
- 0.3 g of an acid is neutralised by 40 $cm^3$ of 0.125 N NaOH. Equivalent mass of the acid is
- Bakelite is manufactured by using a mixture of
- An example for a face centred cubic lattice is
- Coordination number of caesium in caesium chloride is
- Which one of the following is not ferromagnetic?
View More Questions
Top COMEDK UGET General Principles and Processes of Isolation of Elements Questions
- Horn silver is
- Which one of the following ores is obtained by floatation process?
- For the dissociation of limestone in the slag zone of the blast furnace, the reaction is
- Which of the following ores will be concentrated by froth floatation method?
- Liquation process is used for the purification of
View More Questions
Top COMEDK UGET Questions
- If the line px + qy = 0 coincides with one of the lines given by $ax^2 + 2hxy + by^2 = 0$, then
- A long hollow copper pipe carries a current. The magnetic field producted will be
- An ammeter reads 0 to 10 A. It has negligible resistance.To convert this into voltmeter to read upto 250 V, resistance to be used is
- Force of attraction or repulsion between two current carrying wires separated by a distance r is proportional to
- When a material is placed in a magnetic field B, a magnetic moment proportional tc B but in a direction opposite to B is induced. The material is
View More Questions
Concepts Used:
General Principles and Processes of Isolation of Elements
What are Ores and Minerals?
Minerals are the naturally occurring, homogeneous inorganic solid substances. They are having a definite chemical composition and crystalline structure, hardness and color. For example, copper pyrite, calamine, etc.
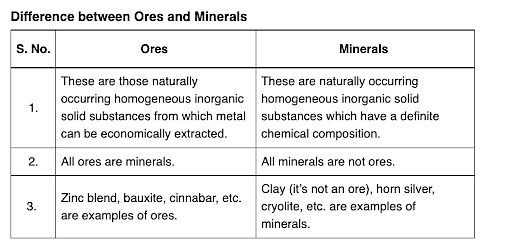
Impurities in an ore are called gauge. The removal of a gauge from the ore is called concentration ore.
Several steps are involved in the extraction of pure metal from ores. Major steps are as follows –
- Concentration of the ore
- Isolation of the metal from its concentrated ore
- Purification of the metal