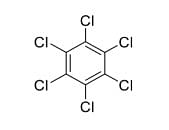
In the following reaction sequence, the major product Q is
\(\text{L-Glucose}\xrightarrow[ii)Cr_2,775K,10-20 \text{atm}]{i)HI, \Delta}P\xrightarrow[UV]{Cl_2(excess)}Q\)
\(\text{L-Glucose}\xrightarrow[ii)Cr_2,775K,10-20 \text{atm}]{i)HI, \Delta}P\xrightarrow[UV]{Cl_2(excess)}Q\)
The Correct Option is D
Solution and Explanation
To solve the problem, we analyze the reaction sequence starting from L-glucose to find the major product Q.
1. Step i) Reaction with HI and heat (\(\Delta\)):
- The sugar undergoes cleavage of C-O bonds by HI.
- This reduces glucose to a mixture of iodoalkanes (polyiodo compounds).
- The carbon skeleton remains but oxygen atoms are replaced by iodine.
2. Step ii) Oxidation with Cr\(_2\)O\(_3\), high temperature and pressure:
- Iodoalkanes are oxidized to form fully chlorinated cyclic hydrocarbons (via halogen exchange).
- Oxygen atoms are removed, and the carbon framework becomes a chlorinated cyclic structure.
3. Step iii) Treatment with excess Cl\(_2\) under UV:
- Further chlorination occurs, leading to a hexachlorinated cyclohexane derivative.
4. Structure of the final product Q:
- Hexachlorocyclohexane (all positions chlorinated).
- This corresponds to structure (D).
Final Answer:
Option (D)
Top JEE Advanced Chemistry Questions
- The molecule that has linear structure is
- Which one among the following does not have the hydrogen bond?
- Carbon tetrachloride has no net dipole moment because of
- On hybridisation of one s and one p-orbital we get
- The IUPAC name of the compound having the formula is
Top JEE Advanced Aldehydes, Ketones and Carboxylic Acids Questions
- When ethanal is treated with Fehling's solution, it gives a precipitate of
- Which of the following compounds will give a yellow precipitate with iodine and alkali ?
- Which of the following will undergo aldol condensation?
- The compound that undergoes decarboxylation most readily under mild condition is
- The carboxyl functional group $(- COOH)$ is present in
Top JEE Advanced Questions
- Yellow light is used in a single slit diffraction experiment with slit width of 0.6 mm. If yellow light is replaced by X-rays, then the observed pattern will reveal
- The size of the image of an object, which is at infinity, as formed by a convex lens of focal length 30 cm is 2 cm. If a concave lens of focal length 20 cm is placed between the convex lens and the image at a distance of 26 cm from the convex lens, calculate the new size of the image.
- A concave lens of glass, refractive index 1.5 has both surfaces of same radius of curvature R. On immersion in a medium of refractive index 1.75, it will behave as a
- The position vector $\vec{r}$ of a particle of mass $m$ is given by the following equation $\vec{r}(t)=\alpha t^{3} \hat{i}+\beta t^{2} \hat{j}$ where $\alpha=\frac{10}{3} \,ms ^{-3}, \beta=5\, ms ^{-2}$ and $m =0.1 \,kg$. At $t =1\, s$, which of the following statement (s) is (are) true about the particle?
- In an n-p-n transistor circuit, the collector current is 10 mA. If 90% of the electrons emitted reach the collector