In a one-litre flask, 6 moles of A undergoes the reaction A (g) ⇌ P (g). The progress of product formation at two temperatures (in Kelvin), T1 and T2, is shown in the figure:

If T1 = 2T2 and (∆G2Θ − ∆G1Θ) = RT2 ln x, then the value of x is ___ .
[∆G1Θ and ∆G2Θ are standard Gibb’s free energy change for the reaction at temperatures T1 and T2, respectively.]

If T1 = 2T2 and (∆G2Θ − ∆G1Θ) = RT2 ln x, then the value of x is ___ .
[∆G1Θ and ∆G2Θ are standard Gibb’s free energy change for the reaction at temperatures T1 and T2, respectively.]
Correct Answer: 8
Solution and Explanation
Gibbs Free Energy Calculation
Given: The chemical equilibrium for the reaction:
\(A(g) \rightleftharpoons P(g)\)
Initially: 6 moles of A and 0 moles of P.
At equilibrium: \(6 - a\) moles of A and \(a\) moles of P.
Solution:
At temperature \( T_1 \), we are given \( a = 4 \). Therefore, the equilibrium constant \( K_{eq1} \) is:
\(K_{eq1} = \frac{4}{2} = 2\)
At temperature \( T_2 \), we are given \( a = 2 \). Therefore, the equilibrium constant \( K_{eq2} \) is:
\(K_{eq2} = \frac{2}{4} = \frac{1}{2}\)
Using the equation for Gibbs free energy change:
\(\Delta G^\circ_1 = -RT \ln(K_{eq1})\)
\(\Delta G^\circ_1 = -2RT \ln(K_{eq1}) \quad \text{[Given: } T_1 = 2T_2 \text{]}\)
\(\Delta G^\circ_2 = -RT \ln(K_{eq2})\)
Now, we subtract the two equations to find the difference in Gibbs free energy:
\(\Delta G^\circ_2 - \Delta G^\circ_1 = RT_2 \ln \left( \frac{(K_{eq1})^2}{K_{eq2}} \right)\)
\(= RT_2 \ln \left( \frac{2^2}{\frac{1}{2}} \right)\)
\(= RT_2 \ln 8\)
Therefore, \( \Delta G^\circ = RT \ln 8 \), and since \( x = 8 \), we conclude that:
The value of \( \Delta G^\circ \) is given by \( RT \ln x \) where \( x = 8 \).
The correct answer is \( x = 8 \).
Top JEE Advanced Chemistry Questions
- The molecule that has linear structure is
- Which one among the following does not have the hydrogen bond?
- Carbon tetrachloride has no net dipole moment because of
- On hybridisation of one s and one p-orbital we get
- The IUPAC name of the compound having the formula is
Top JEE Advanced Law Of Chemical Equilibrium And Equilibrium Constant Questions
- The precipitate of C$aF_2, \, ( K_{sp} = 1.7 \times 10^{ - 10 } )$ is obtained, when equal volumes of which of the following are mixed?
- In a one-litre flask, 6 moles of A undergoes the reaction A (g) ⇌ P (g). The progress of product formation at two temperatures (in Kelvin), T1 and T2, is shown in the figure:

If T1 = 2T2 and (∆G2Θ − ∆G1Θ) = RT2 ln x, then the value of x is ___ . [∆G1Θ and ∆G2Θ are standard Gibb’s free energy change for the reaction at temperatures T1 and T2, respectively.]
- For the reaction, $X (s) \rightleftharpoons Y (s)+ Z (g)$, the plot of $\ln \frac{p_{ Z }}{p^{0}}$ versus $\frac{10^{4}}{T}$ is given below (in solid line), where $p_{ z }$ is the pressure (in bar) of the gas $Z$ at temperature $T$ and $p^0=1$ bar
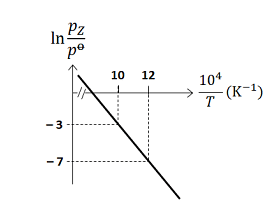
(Given, $\frac{ d (\ln K)}{ d \left(\frac{1}{T}\right)}=-\frac{\Delta H^{0}}{R}$, where the equilibrium constant, $K=\frac{p_{z}}{p^{0}}$ and the gas constant, $R=8314 \,J \,K ^{-1} mol ^{-1}$ )
The value of standard enthalpy, $\Delta H^{0}$ (in $kJ\, mol ^{-1}$ ) for the given reaction is ______ - For the reaction, $X (s) \rightleftharpoons Y (s)+ Z (g)$, the plot of $\ln \frac{p_{ Z }}{p^{0}}$ versus $\frac{10^{4}}{T}$ is given below (in solid line), where $p_{ z }$ is the pressure (in bar) of the gas $Z$ at temperature $T$ and $p^0=1$ bar
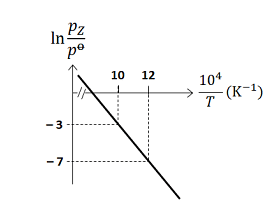
(Given, $\frac{ d (\ln K)}{ d \left(\frac{1}{T}\right)}=-\frac{\Delta H^{0}}{R}$, where the equilibrium constant, $K=\frac{p_{z}}{p^{0}}$ and the gas constant, $R=8.314 \,J \,K ^{-1} mol ^{-1}$ )
The value of $\Delta S^{0}$ (in $J\, K ^{-1} mol ^{-1}$ ) for the given reaction, at $1000\, K$ is ______ - For the reaction, $X (s) \rightleftharpoons Y (s)+ Z (g)$, the plot of $\ln \frac{p_{ Z }}{p^{0}}$ versus $\frac{10^{4}}{T}$ is given below (in solid line), where $p_{ z }$ is the pressure (in bar) of the gas $Z$ at temperature $T$ and $p^0=1$ bar
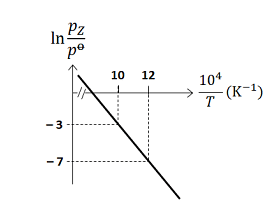
(Given, $\frac{ d (\ln K)}{ d \left(\frac{1}{T}\right)}=-\frac{\Delta H^{0}}{R}$, where the equilibrium constant, $K=\frac{p_{z}}{p^{0}}$ and the gas constant, $R=8314 \,J \,K ^{-1} mol ^{-1}$ )
Top JEE Advanced Questions
- Yellow light is used in a single slit diffraction experiment with slit width of 0.6 mm. If yellow light is replaced by X-rays, then the observed pattern will reveal
- The size of the image of an object, which is at infinity, as formed by a convex lens of focal length 30 cm is 2 cm. If a concave lens of focal length 20 cm is placed between the convex lens and the image at a distance of 26 cm from the convex lens, calculate the new size of the image.
- A concave lens of glass, refractive index 1.5 has both surfaces of same radius of curvature R. On immersion in a medium of refractive index 1.75, it will behave as a
- The position vector $\vec{r}$ of a particle of mass $m$ is given by the following equation $\vec{r}(t)=\alpha t^{3} \hat{i}+\beta t^{2} \hat{j}$ where $\alpha=\frac{10}{3} \,ms ^{-3}, \beta=5\, ms ^{-2}$ and $m =0.1 \,kg$. At $t =1\, s$, which of the following statement (s) is (are) true about the particle?
- In an n-p-n transistor circuit, the collector current is 10 mA. If 90% of the electrons emitted reach the collector
Concepts Used:
Law of Chemical Equilibrium
Law of Chemical Equilibrium states that at a constant temperature, the rate of a chemical reaction is directly proportional to the product of the molar concentrations of the reactants each raised to a power equal to the corresponding stoichiometric coefficients as represented by the balanced chemical equation.
Let us consider a general reversible reaction;
A+B ↔ C+D
After some time, there is a reduction in reactants A and B and an accumulation of the products C and D. As a result, the rate of the forward reaction decreases and that of backward reaction increases.
Eventually, the two reactions occur at the same rate and a state of equilibrium is attained.
By applying the Law of Mass Action;
The rate of forward reaction;
Rf = Kf [A]a [B]b
The rate of backward reaction;
Rb = Kb [C]c [D]d
Where,
[A], [B], [C] and [D] are the concentrations of A, B, C and D at equilibrium respectively.
a, b, c, and d are the stoichiometric coefficients of A, B, C and D respectively.
Kf and Kb are the rate constants of forward and backward reactions.
However, at equilibrium,
Rate of forward reaction = Rate of backward reaction.
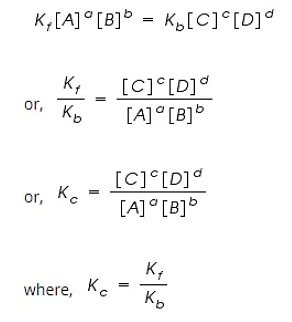
Kc is called the equilibrium constant expressed in terms of molar concentrations.
The above equation is known as the equation of Law of Chemical Equilibrium.