Following reaction can produce three products. The order of their amount is
Following reaction can produce three products. The order of their amount is
A> C> B
C> B> A
A> B> C
C> A> B
The Correct Option is A
Solution and Explanation
The correct option is (A): A> C> B
Top SRMJEEE Chemistry Questions
How many enolizable hydrogens are there in the compound 2-methylcyclohex-2- en -1 -one?
When the valence d orbitals of the central metal ion in tetrahedral complex are split in energy levels in CFT, which orbitals are raised to higher energy?
Which of the following statement is true for aqueous solution of 0.1 M urea, 0.2 M glucose nad 0.3 M sucrose
The hybridization of nitrogen in diethyl amine is
Amino acids generally exist in the form of Zwitter ions.
Top SRMJEEE Law Of Chemical Equilibrium And Equilibrium Constant Questions
Top SRMJEEE Questions
- Three charges -q, Q and -q are placed in a straight line maintaining equal distance from each other. What should be the ratio \(\frac{q}{Q}\) so that net electric potential of the system is zero?
- What can be said regarding a line if its slope is negative?
- An ideal diode presents a(n)______ when reversed-biased and a(n) __________ when forward-biased.
How many enolizable hydrogens are there in the compound 2-methylcyclohex-2- en -1 -one?
When the valence d orbitals of the central metal ion in tetrahedral complex are split in energy levels in CFT, which orbitals are raised to higher energy?
Concepts Used:
Law of Chemical Equilibrium
Law of Chemical Equilibrium states that at a constant temperature, the rate of a chemical reaction is directly proportional to the product of the molar concentrations of the reactants each raised to a power equal to the corresponding stoichiometric coefficients as represented by the balanced chemical equation.
Let us consider a general reversible reaction;
A+B ↔ C+D
After some time, there is a reduction in reactants A and B and an accumulation of the products C and D. As a result, the rate of the forward reaction decreases and that of backward reaction increases.
Eventually, the two reactions occur at the same rate and a state of equilibrium is attained.
By applying the Law of Mass Action;
The rate of forward reaction;
Rf = Kf [A]a [B]b
The rate of backward reaction;
Rb = Kb [C]c [D]d
Where,
[A], [B], [C] and [D] are the concentrations of A, B, C and D at equilibrium respectively.
a, b, c, and d are the stoichiometric coefficients of A, B, C and D respectively.
Kf and Kb are the rate constants of forward and backward reactions.
However, at equilibrium,
Rate of forward reaction = Rate of backward reaction.
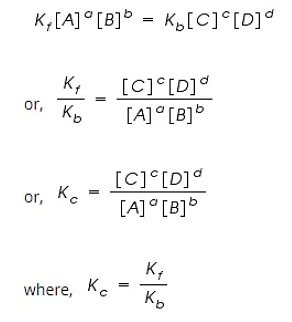
Kc is called the equilibrium constant expressed in terms of molar concentrations.
The above equation is known as the equation of Law of Chemical Equilibrium.