Based on the given figure, the number of correct statement/s is/are _______
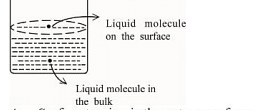
A. Surface tension is the outcome of equal attractive and repulsive forces acting on the liquid molecule in bulk
B. Surface tension is due to uneven forces acting on the molecules present on the surface
C. The molecule in the bulk can never come to the liquid surface
D. The molecules on the surface are responsible for vapour pressure if the system is a closed system

A. Surface tension is the outcome of equal attractive and repulsive forces acting on the liquid molecule in bulk
B. Surface tension is due to uneven forces acting on the molecules present on the surface
C. The molecule in the bulk can never come to the liquid surface
D. The molecules on the surface are responsible for vapour pressure if the system is a closed system
Correct Answer: 2
Solution and Explanation
The correct options are:
B: Surface tension arises due to uneven forces acting on the molecules present at the surface. Molecules at the surface of a liquid experience a net inward force because they are not surrounded by similar molecules on all sides. This imbalance results in the liquid's surface behaving as if it were under tension.
D: The molecules at the surface are responsible for vapor pressure in a closed system. In a closed system, molecules at the surface of the liquid are able to escape into the vapor phase, contributing to the vapor pressure. The equilibrium between the liquid and vapor phases determines the vapor pressure.
Top JEE Main Chemistry Questions
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- At equilibrium the concentrations of and in a sealed vessel at . What will be for the reaction
- Find out the major products from the following reactions

Cobalt chloride when dissolved in water forms pink colored complex $X$ which has octahedral geometry. This solution on treating with cone $HCl$ forms deep blue complex, $\underline{Y}$ which has a $\underline{Z}$ geometry $X, Y$ and $Z$, respectively, are
- The correct order of atomic radii is:
Top JEE Main States of matter Questions
- An open vessel at $27^{\circ}C$ is heated until two fifth of the air (assumed as an ideal gas) in it has escaped from the vessel. Assuming that the volume of the vessel remains constant, the temperature at which the vessel has been heated is :
- Points I, II and III in the following plot respectively correspond to ($V_{mp} :$ most probable velocity)
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.
Concepts Used:
States of Matter
The matter is made up of very tiny particles and these particles are so small that we cannot see them with naked eyes.
There are three States of Matter:
The three states of matter are as follows:
Solid State:
- The solid-state is one of the fundamental states of matter.
- Solids differ from liquids and gases by the characteristic of rigidity.
- The molecules of solids are tightly packed because of strong intermolecular forces; they only oscillate about their mean positions.
Liquid State:
- The molecules in a liquid are closely packed due to weak intermolecular forces.
- These forces are weaker than solids but stronger than that of gases.
- There is much space in between the molecules of liquids which makes their flowing ability easy.
Gaseous State:
- In this state of matter, distances between the molecules are large (intermolecular distance is in the range of 10-7-10-5 cm.
- The intermolecular forces experienced between them are negligible.
- Thus, translatory, rotatory and vibratory motions are observed prominently in gases.