Question:
A reversible heat engine absorbs 20 kJ of heat from a source at 500 K and dissipates it to the reservoir at 400 K. The efficiency of the heat engine is \(\underline{\hspace{2cm}}\) %.
A reversible heat engine absorbs 20 kJ of heat from a source at 500 K and dissipates it to the reservoir at 400 K. The efficiency of the heat engine is \(\underline{\hspace{2cm}}\) %.
Show Hint
The efficiency of a Carnot engine depends on the temperatures of the heat source and sink. The higher the temperature difference, the higher the efficiency.
Updated On: Jan 7, 2026
Show Solution
Verified By Collegedunia
Correct Answer: 20
Solution and Explanation
The efficiency of a reversible heat engine (Carnot engine) is given by the formula:
\[ \eta = 1 - \frac{T_C}{T_H} \] where \( T_H \) is the temperature of the heat source and \( T_C \) is the temperature of the heat sink.
Here, \( T_H = 500 \, K \) and \( T_C = 400 \, K \). Substituting the values into the formula:
\[ \eta = 1 - \frac{400}{500} = 1 - 0.8 = 0.2 \] Thus, the efficiency of the heat engine is \( 20% \).
\[ \eta = 1 - \frac{T_C}{T_H} \] where \( T_H \) is the temperature of the heat source and \( T_C \) is the temperature of the heat sink.
Here, \( T_H = 500 \, K \) and \( T_C = 400 \, K \). Substituting the values into the formula:
\[ \eta = 1 - \frac{400}{500} = 1 - 0.8 = 0.2 \] Thus, the efficiency of the heat engine is \( 20% \).
Was this answer helpful?
0
0
Top GATE CY Organic Chemistry Questions
- The major product in the following reaction sequence is
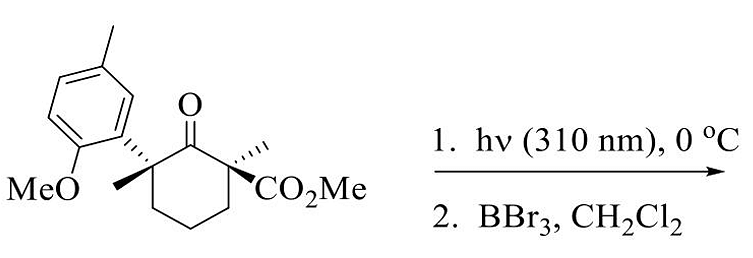
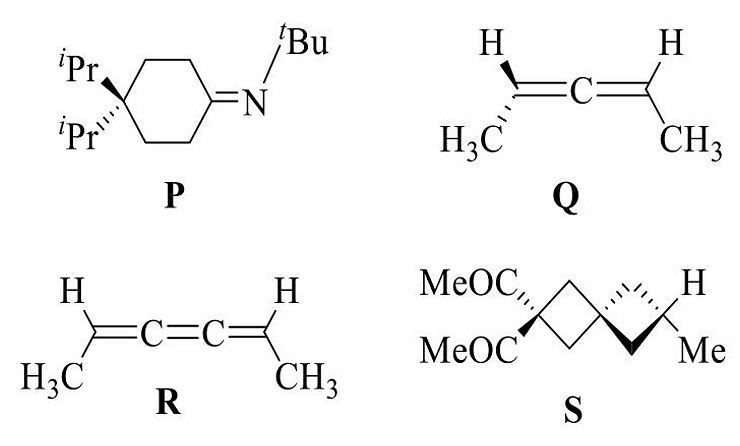
- Among the following, the chiral compound is
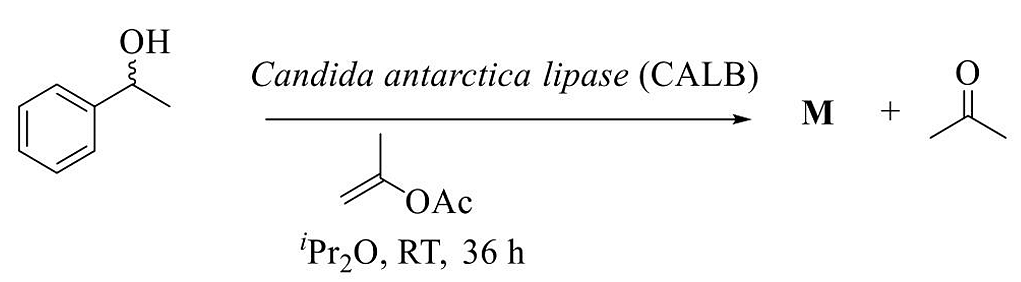
- The product M in the following reaction is
- The reaction(s) that yield(s) X as the major product is (are)
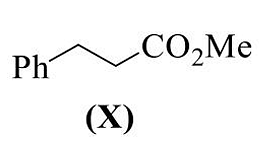
- The reaction(s) that yield(s) 2-methylquinoline as the major product is (are)
View More Questions
Top GATE CY Questions
- Ψ(x) = \(\sqrt{\frac{32}{L^3}}\)[sin(\(\frac{\pi x}{L}\)).cos(\(\frac{\pi x}{L}\)).sin(\(\frac{2\pi y}{L}\)).sin(\(\frac{\pi z}{L}\))]Find the value of E221: E111.
 what is the final product
what is the final product- Which are optically active?
 intensity ratio of final product
intensity ratio of final product- Which among the following is true?
View More Questions