17 mg of a hydrocarbon (M.F. C10H16) takes up 8.40 mL of the H2 gas measured at \(0\degree C\) and 760 mm of Hg. Ozonolysis of the same hydrocarbon yields

The number of double bond/s present in the hydrocarbon is_______.

The number of double bond/s present in the hydrocarbon is_______.
Show Hint
For determining double bonds:
• Use the degree of unsaturation formula to find the total number of double bonds and rings.
• Analyze ozonolysis fragments to confirm the number and position of double bonds.
Correct Answer: 3
Solution and Explanation
1. Molecular Formula Analysis:
The given molecular formula is \(\text{C}_{10}\text{H}_{16}\). The degree of unsaturation is:
\[\text{Degree of unsaturation} = \frac{2C + 2 - H}{2} = \frac{2(10) + 2 - 16}{2} = 3.\]
This indicates 3 double bonds or rings.
2. Hydrogenation Data:
8.40 mL of \(\text{H}_2\) gas is absorbed. At STP, 1 mol of gas occupies 22.4 L. Moles of \(\text{H}_2\) absorbed:
\[n = \frac{8.40}{22400} = 3.75 \times 10^{-4}~\text{mol}.\]
3. Ozonolysis Data:
Ozonolysis fragments indicate three distinct double bonds in the hydrocarbon.
4. Conclusion
The hydrocarbon contains 3 double bonds.
Final Answer: \(3\).
Top JEE Main Chemistry Questions
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- At equilibrium the concentrations of and in a sealed vessel at . What will be for the reaction
- Find out the major products from the following reactions

Cobalt chloride when dissolved in water forms pink colored complex $X$ which has octahedral geometry. This solution on treating with cone $HCl$ forms deep blue complex, $\underline{Y}$ which has a $\underline{Z}$ geometry $X, Y$ and $Z$, respectively, are
- The correct order of atomic radii is:
Top JEE Main Hydrocarbons Questions
- A \(10\) \(mL\) hydrocarbon \((C_2H_4)\) on combustion give \(40\) \(mL\) \(CO_2\) and \(50\) \(mL\) \(H_20\). Calculate the value of \(x+y\)
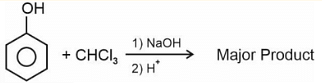
The major product in the above reaction is- 3-Methylhex-2-ene on reaction with HBr in presence of peroxide forms an addition product (A). The number of possible stereoisomers for 'A' is ______.
- Cyclohexene
 is _________ type of an organic compound.
is _________ type of an organic compound. - Find product P of the following reaction-
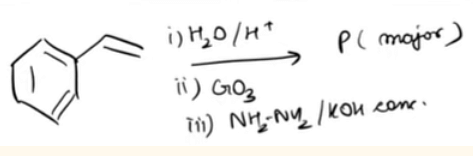
Top JEE Main Questions
- In a hydrogen like atom, when an electron jumps from the $M$ - shell to the $L$ - shell, the wavelength of emitted radiation is $\lambda$. If an electron jumps from $N$-shell to the $L$-shell, the wavelength of emitted radiation will be :
- Two masses $m$ and $\frac{m}{2}$ are connected at the two ends of a massless rigid rod of length $l$. The rod is suspended by a thin wire of torsional constant $k$ at the centre of mass of the rod-mass system(see figure). Because of torsional constant $k$, the restoring torque is $\tau =k\theta$ for angular displacement $\theta$. If the rod is rota ted by $\theta_0$ and released, the tension in it when it passes through its mean position will be:
What will be the equilibrium constant of the given reaction carried out in a \(5 \,L\) vessel and having equilibrium amounts of \(A_2\) and \(A\) as \(0.5\) mole and \(2 \times 10^{-6}\) mole respectively?
The reaction : \(A_2 \rightleftharpoons 2A\)- The value of is
- The number of terms of an is even. The sum of the odd terms is and of the even terms is . The last term exceeds the first by . Then the number of terms in the series is ______.